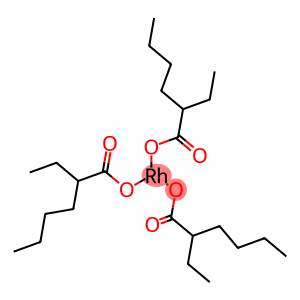
Tris(2-ethylhexanoate)rhodiuM (III)
Tris(2-ethylhexanoate)rhodium (III) - solution
CAS: 20845-92-5
Molecular Formula: C24H45O6Rh
Tris(2-ethylhexanoate)rhodiuM (III) - Names and Identifiers
Tris(2-ethylhexanoate)rhodiuM (III) - Physico-chemical Properties
| Molecular Formula | C24H45O6Rh |
| Molar Mass | 532.52 |
| Boling Point | 228°C at 760 mmHg |
| Flash Point | 116.6°C |
| Water Solubility | 9.8mg/L |
| Vapor Presure | 0.027mmHg at 25°C |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
Tris(2-ethylhexanoate)rhodiuM (III) - Reference Information
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Application | Rhodium 2-ethylhexanoate is an important noble metal compound commonly used in the chemical and electronic industries. Due to its high solubility in various organic solvents, large rhodium content and strong stability, it has been widely used in the manufacture of precious metal thin film layer coatings for solar cells. |
| preparation | a simple, efficient, energy-saving and environmentally friendly process for the production of rhodium 2-ethylhexanoate. In the kettle, 210g(1.456 moles) of 2-ethylhexanoic acid and 12.7g of deionized water and 0.317g (moles) of sodium hydroxide were added. Stir at 60 °c for 1 hour. 10g(0.048 mol) of rhodium trichloride was added. The hot water was discharged and heated to 100 degrees Celsius with steam and refluxed for 24 hours. After completion of the reaction, the temperature was cooled to room temperature, and the mixture was allowed to stand and layered. 1N aqueous sodium hydroxide solution was added dropwise to the oil phase at 30-50 ° C. To adjust the pH of the reaction system to pH = 6-10. After stirring for 20 minutes, the pH was stabilized, the mixture was washed with an appropriate amount of deionized water, stirred for 20 minutes, and then allowed to stand for 18 hours, suction filtered, and the crystals were washed with deionized water and dried. It was dried at 100 °c under vacuum and the filtrate was drained. Yield 99%, purity of liquid phase: 98.5%. |
Last Update:2024-04-09 15:16:45
Supplier List
Product Name: Rhodium tris(2-ethylhexanoate) Request for quotation
CAS: 20845-92-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 20845-92-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: Rhodium tris(2-ethylhexanoate) Request for quotation
CAS: 20845-92-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 20845-92-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
View History
